
An unmissable update from Ronny Allan covering April 2026
A monthly update not to miss……..Summary of April 2026 on RonnyAllan.NET I think the main story of April is similar to March which was a strong
The FDA Fast Track is a process designed to facilitate the development and expedite the review of drugs to treat serious conditions and address an unmet medical need. The purpose is to bring new and promising medicines to patients sooner. The Fast Track designation enables ITM to have more frequent interactions with the FDA to discuss the ITM-11 development path. It also allows rolling review of the new drug application (NDA) for ITM-11, when submitted. The rolling submission will allow ITM to submit completed sections of an application for review by FDA, rather than wait until all sections are completed.
COMPETE – low grade 1 and 2 NET
COMPOSE – aggressive grade 2 and 3 NET
Update 22 Jan 2022.
ITM Isotope Technologies Munich SE (ITM), today announced that the first patient has been treated in its second pivotal phase III clinical trial, COMPOSE (NCT04919226), evaluating the company’s lead radiopharmaceutical candidate, ITM-11 (n.c.a. 177Lu-edotreotide), for the treatment of gastroenteropancreatic neuroendocrine tumors (GEP-NETs). ITM-11 is a Targeted Radionuclide Therapeutic consisting of the high-quality radioisotope no-carrier-added lutetium-177 (n.c.a. 177Lu) fused with a somatostatin analogue to specifically target somatostatin receptor-positive (SSTR+) GEP-NETs.
The trial design of COMPOSE was presented at the 2022 ASCO Gastrointestinal Cancers Symposium (ASCO-GI).
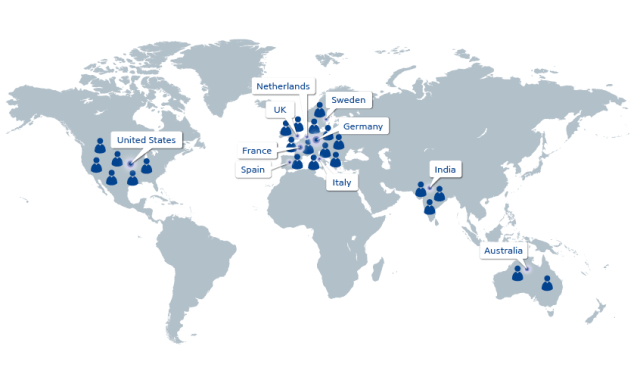
Graphic courtesy of ITM – clinical trial sites as of 14 Oct 2021
This is an Open-label, Multicentre Study. Its purpose is to evaluate the efficacy, safety & patient-reported outcomes of peptide receptor radionuclide therapy (PRRT) with 177Lu-Edotreotide as 1st or 2nd line of treatment compared to best standard of care in patients with well-differentiated aggressive grade 2 and grade 3, somatostatin receptor-positive (SSTR+), neuroendocrine tumours of gastroenteric or pancreatic origin. The standard of care comparison is based on either CAPTEM or everolimus or FOLFOX in patients with advanced grade 2 or grade 3 GEP-NETs.
So, this trial is focused on more aggressive tumours, which is great news.
1. ITM Website – click here
2. COMPOSE Clinical Trials Document – click here
3. COMPOSE Phase 2 results – click here
click here or on the picture

While COMPOSE is evaluating ITM-11 for the treatment of well-differentiated high (ki67) grade 2 and grade 3 GEP-NETs, the radiopharmaceutical is also being investigated in ITM’s ongoing pivotal phase III trial, COMPETE (NCT03049189), in patients with grade 1 and low (ki67) grade 2 GEP-NETs.
I’ve written about this product before. It’s a synthetic, low-energy beta-emitting isotope of Lutetium 177 (a radioactive component already used within an approved product called Lutathera). Attaching the radiopharmaceutical Solucin® it’s administered as an intravenous infusion, specifically targeting and destroying the tumor cells with ionizing radiation, i.e. it works in exactly the same way as conventional PRRT. Read more by clicking this link plus news of an existing trial in Phase 3 – Phase 3 Clinical Trial of 177Lu-Edotreotide (Solucin®) – COMPETE for GEPNETs
Choosing to participate in a study is an important personal decision. Talk with your doctor and family members or friends about deciding to join a study. To learn more about this study, you or your doctor may contact the study research staff using the contacts provided in the clinical trials document. It’s very important to check the trial inclusion and exclusion criteria before making any contact. If you need questions, the articles here is very useful Questions to Ask About Clinical Trials | Cancer.Net
The inclusion of any trial within this blog should not be taken as a recommendation by Ronny Allan.
I am not a doctor or any form of medical professional, practitioner or counsellor. None of the information on my website, or linked to my website(s), or conveyed by me on any social media or presentation, should be interpreted as medical advice given or advised by me.
Neither should any post or comment made by a follower or member of my private group be assumed to be medical advice, even if that person is a healthcare professional. Some content may be generated by AI which can sometimes be misinterpreted. Please check any references attached.
Please also note that mention of a clinical service, trial/study or therapy does not constitute an endorsement of that service, trial/study or therapy by Ronny Allan, the information is provided for education and awareness purposes and/or related to Ronny Allan’s own patient experience. This element of the disclaimer includes any complementary medicine, non-prescription over the counter drugs and supplements such as vitamins and minerals.
Whenever I post about a trial or study, some people get excited without understanding that these new treatments and capabilities can very often take years to come to fruition and it’s also possible that clinical trials can be halted, or that national approval agencies will not approve the final product. Plus, not everyone will be eligible, so always check the exclusion and inclusion criteria in the relevant clinical trials document. Please bear that in mind when reading studies/clinical trials posted on RonnyAllan.NET

A monthly update not to miss……..Summary of April 2026 on RonnyAllan.NET I think the main story of April is similar to March which was a strong

DisclaimerThis Spotlight is for general education and reassurance only. It cannot replace personalised advice from your own medical team, who understand your individual history, imaging,

DisclaimerThe information in this Spotlight is for general education only. It cannot replace advice from your own medical team, who know your individual situation, test

I was delighted to read about the efforts of Dr Arthur Scott, a Member of the UK Parliament (MP). He is a great advocate for

DisclaimerThis information is for education and reassurance only.It is not a substitute for personalised medical advice, diagnosis, or treatment. Bone metastases in neuroendocrine tumours (NETs)

Before you read thisThis information is designed to help you understand how vitamins work in the body and how certain NET-related factors might affect them.

Here is my monthly summary of March 2026 on RonnyAllan.NET This has been a very good month but to be honest, I had more time at

Before you read this… This article discusses pathology concepts such as Ki-67, grading, heterogeneity, and biopsy findings in neuroendocrine tumours (NETs). It is provided for

Before you read this… This article is provided to support understanding of a complex and evolving topic. It explains how liver transplant is being explored
I would also mention those who contributed to my “Tea Fund” which resides on PayPal. You don’t need a PayPal account as you can select a card but don’t forget to select the number of units first (i.e. 1 = £4, 2 = £8, 3 = £12, and so on), plus further on, tick a button to NOT create a PayPal account if you don’t need one. Clearly, if you have a PayPal account, the process is much simpler
Through your generosity, I am able to keep my sites running and provide various services for you. I have some ideas for 2023 but they are not detailed enough to make announcements yet.
This screenshot is from every single post on my website and depending on which machine you are using, it will either be top right of the post or at the bottom (my posts are often long, so scroll down!)
Subscribe to get the latest posts sent to your email.
Subscribe now to keep reading and get access to the full archive.